mod education are provide best solution for NCERT solutions for Class 8 Science Chapter 6 Combustion and Flame Notes and Que Answer and Video Class.
NCERT Solutions for Class 8 Science Chapter 6 Combustion and Flame Notes
| Class | 8th Class |
| Chapter | Chapter 6 |
| Chapter Name | Combustion and Flame |
| Language | English |
| Book | Science |
We use different kinds of fuel for various purposes at home, in industry and for running automobiles. These fuels are cow dung, wood, coal, charcoal, petrol, diesel, compressed natural gas (CNG) etc.
Combustion: A chemical process in which a substance reacts with oxygen to give off heat is called combustion.
Fuel or Combustible Substance: Any material that undergoes combustion is called a combustible substance. It is also called as fuel. Some examples of fuels are petrol, diesel, etc. The fuel may be solid, liquid or gas. Sometimes, light is also given off during combustion, either as a flame or as a glow.
Ignition Temperature:
- It is the lowest temperature at which a substance catches fire.
- If the temperature of a combustible substance is lower than the ignition temperature, then the substance will not burn.
- Example: (i)Cooking oil catches fire when a frying pan is kept for long on a burning stove.
(ii) Kerosene oil and wood do not catch fire on their own at room temperature. But, if kerosene oil is heated a little, it will catch fire. But if wood is heated a little, it would still not catch fire.
Inflammable Substances: Those substances which have very low ignition temperature and can easily catch fire with a flame are termed as inflammable substances. Examples: petrol, alcohol, LPG, etc.
Conditions necessary for combustion to take place:
- Fuel or Combustible substance
- Air (With presence of Oxygen in it)
- Heat (to raise the temperature of the fuel beyond the ignition temperature)
Forest Fires: In summer season, when temperature rises too high, then the regions having dry grass will catch the fire. This fire spreads rapidly from grass to trees and eventually entire forest is on fire. And it is difficult to manage such fires.
Types of Combustion:
- Rapid Combustion
In this type of combustion, the substances burns rapidly and yield light and heat. Example: Bring a burning matchstick or a gas lighter near a gas stove in the kitchen. Turn on the knob of the gas stove. We find that the gas burns rapidly and produces heat and light. - Spontaneous Combustion
In this type of combustion, substances burst out into flames suddenly without the application of any apparent cause. Examples: Many disastrous fires in coal mines result due to this kind of combustion. Spontaneous forest fires are sometimes due to the heat of the sun or due to lightning strike. - Explosion
In this type of combustion, all of a sudden reaction results into heat, light and sound. Moreover, large quantity of gas also gets released. Example: When a fire cracker is ignited, a sudden reaction takes place with the evolution of heat, light and sound with the large amount of gas.
Flame: When something is burnt, a hot luminous gas emerges out of the substance. This gas is called as flame.
The substances which vaporise during burning, give flames. For example, kerosene oil and molten wax rise through the wick and are vapourised during burning and form flames. Charcoal, on the other hand, does not vapourise and so does not produce a flame.
NCERT Solutions Class 8 Science Chapter 6 Que & Ans
1. List conditions under which combustion can take place.
Answer: Combustion is burning of substance in presence of oxygen. Following are the conditions under which combustion takes place –
- Presence of oxygen
- Presence of fuel
- Ignition temperature
2. Fill in the blanks. NCERT Solutions for Class 8 Science Chapter 6
(a) Burning of wood and coal causes _________of air.
Answer: pollution
(b) A liquid fuel, used in homes is ___________.
Answer: liquefied petroleum gas
(c) Fuel must be heated to its__________ _________ before it starts burning.
Answer: ignition temperature
(d) Fire produced by oil cannot be controlled by _________.
Answer: water
3. Explain how the use of CNG in automobiles has reduced pollution in our cities.
Answer: Combustion of petroleum produces carbon monoxide gas. It causes respiratory disease. Use of CNG in automobiles produces very less amount of harmful product. Thus, it helps in reducing pollution.
4. Compare LPG and wood as fuels.
Answer: Wood is used as tradition method of fuel. Wood causes pollution by producing smoke. Also, wood is obtained from trees, thus, using wood as fuel will involve cutting of tree, which is bad for environment. If LPG is used as a fuel, it does not produce smoke or other harmful pollutants. Moreover, LPG is more efficient than wood in terms of heat production.
5. Give reasons.
(a) Water is not used to control fires involving electrical equipment.
Answer: Water is not used for controlling fires involving electrical equipment because water is good conductor electricity. Person controlling fire with water will get a shock or it can damage electrical equipment. Also it can transfer current in all areas through water.
(b) LPG is a better domestic fuel than wood.
Answer: LPG produces very less smoke or other harmful pollutants in comparison to wood. Moreover, LPG is more efficient than wood in terms of heat production.
(c) Paper by itself catches fire easily whereas a piece of paper wrapped around an aluminium pipe does not.
Answer: Paper wrapped around an aluminium pipe does not catches fire because heat absorbed by paper is transferred to aluminium. Aluminium being metal is good conductor. Thus heat get transferred to aluminium and paper does not reach at its ignition temperature.
6. Make a labelled diagram of a candle flame.
Answer:
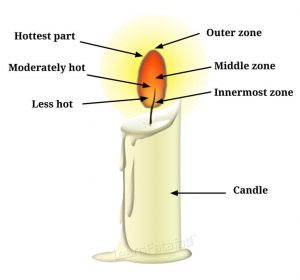
7. Name the unit in which the calorific value of a fuel is expressed.
Answer: CO2 is heavier than oxygen, when sprayed on fire it breaks the contact of fuel and oxygen, thereby controlling fire. In cylinders, carbon dioxide is stored in liquid form, while extinguishing fire, it expands and cools down and lowers the temperature of fuel. Thus, it controls fires.
9. It is difficult to burn a heap of green leaves but dry leaves catch fire easily. Explain.
Answer: Green leaves has presence of moisture in them, this do not allow green leaves to reach its ignition temperature and thus it prevents burning. Conversely, in dry leaves moisture is absent, therefore, it easily reaches at ignition temperature thereby catching fire easily.
10. Which zone of a flame does a goldsmith use for melting gold and silver and why?
Answer: Goldsmith uses outer flame to melt gold and silver, because it is the hottest part of the flame and undergoes complete combustion.
11. In an experiment 4.5 kg of a fuel was completely burnt. The heat produced was measured to be 180,000 kJ. Calculate the calorific value of the fuel.
Answer: Given: 4.5 kg fuel produces 180000 kilojules of heat.
∴ Heat produced by 1 kg of fuel = ( 180000 / 4.5 ) x 1 kJ /kg = 40000 kJ/kg
12. Can the process of rusting be called combustion? Discuss.
Answer: Combustion is chemical process in which substance reacts with oxygen and produces energy to give heat and light. In rusting, exothermic process is carried out which releases heat, thus, rusting can be named as slow combustion.
13. Abida and Ramesh were doing an experiment in which water was to be heated in a beaker. Abida kept the beaker near the wick in the yellow part of the candle flame. Ramesh kept the beaker in the outermost part of the flame. Whose water will get heated in a shorter time?
Answer: Water in Ramesh’s beaker will get heated in less time, as he has kept beaker in outermost part of the flame, which is the hottest part. Water from Abida beaker will get heated in longer time as she has kept beaker near the wick in the yellow part of the candle flame which is less hot than outer part of flame.
NCERT Solutions Class 8 Science Chapter 6 Shorts and MCQS
Question 1. Name the most common fuel used in homes.
Answer: Liquefied Petroleum Gas. (LPG)
Question 2. Name the most common fire extinguisher.
Answer: Water
Question 3. What are the states in which a fuel may exist?
Answer: A fual may exist in solid, liquid or gaseous state.
Question 4. ame any two combustible substances.
Answer: Charcoal, wood
Question 5. Does magnesium produce heat and light during its combustion?
Answer: Yes, it does.
Question 6. What acts as a fuel for our body?
Answer: Food
Question 7. Give two examples of non-combustible substances.
Answer: Water, sand
Question 8. How are heat and light produced in the sun?
Answer: In the sun, heat and light are produced by nuclear reactions.
Question 9. Where were matchsticks first used?
Answer: Egypt
Question 10. What are the three essential requirements for combustion?
Answer: Fuel, air (to supply oxygen) and heat (to raise the temperature of the fuel beyond the ignition temperature).
Question 11. What is the ignition temperature of phosphorus?
Answer: 25°C
Question 12. What is the colour of an LPG flame?
Answer: Blue
Question 13. What type of process is combustion?
Answer: A chemical process
Question 14. Name an ideal fuel.
Answer: Compressed Natural Gas (CNG)
Question 15. What is the composition of the head of a matchstick?
Answer: Antimony trisulphide and potassium chlorate.
Question 16. Which part of a flame does a goldsmith blow for melting gold and silver?
Answer: The goldsmith blows the outermost zone of a flame for melting gold and silver.
Question 17. What is the unit for expressing the calorific value of a fuel?
Answer: Kilojoules per kilogram (kJ/kg)
Question 18. Comparing the calorific values of coal and petrol, state which fuel is better.
Answer: The calorific value of coal is about 25,000 – 33,000 kJ/kg, whereas that of petrol is 45,000 kJ/kg. Hence, petrol is better tuel.
Question 19. What is deforestation?
Answer: The cutting down of trees on a large scale is termed as deforestation.
Question 20. Give any two examples of carbon fuels.
Answer: Coal, petroleum
