NCERT Solutions, Question Answer and Mind Map for Class 12 Chemistry Chapter 16, “Chemistry in Everyday Life,” is a study material package designed to help students understand the role of chemistry in everyday life, the different types of drugs and medicines, their classification, and the principles behind their actions.
NCERT Solutions provide detailed explanations and answers to the questions presented in the chapter. The solutions cover all the topics in the chapter, including the classification of drugs, the principles of drug action, and the use of artificial sweeteners, antioxidants, and food preservatives. They also provide tips on how to answer different types of questions, including short answer, long answer, and multiple-choice questions.

The question-answer section of the chapter covers a wide range of topics, from the different types of drugs and their classifications to the principles of drug action, including the mechanism of drug-receptor interactions. It also includes questions on the use of food additives and the health hazards associated with their use.
The mind map provides a visual representation of the key topics covered in the chapter, allowing students to understand the connections between different concepts and ideas. The mind map covers the classification of drugs, their mechanism of action, and the use of food additives and preservatives.
NCERT Solution / Notes Class 12 Chemistry Chapter 16 Chemistry in Everyday Life with Mind Map PDF Download
CHEMISTRY IN EVERYDAY LIFE
Introduction: In this Unit, we shall learn the application of Chemistry in three important and interesting areas, namely–medicines, food materials and cleansing agents. For cleanliness, we use soaps, detergents, toothpaste, bleaches etc., which all are made up of chemical compounds.
Similarly, Clothes (Cotton, Wool, Silk, Terylene), Food materials (Carbohydrates, Proteins, Oil, Fats), Medicines (Antibiotics, Antimalarials etc.), Explosives, Fuels, Rocket propellants, Building materials etc. are all chemical compounds or derived from them.
Importance of Chemistry in our daily life: Medicines
A drug is a chemical agent, which affects human metabolism and provides cure from ailment.
Or Drugs are chemicals which interact with macromolecular targets and produce a biological response.
Drugs will interact with macromolecules targets (where these targets are chemical compounds like proteins, lipids etc.) in order to maintain balance in the body.
When the biological response is therapeutic and useful, these chemicals are called medicines and are used in diagnosis, prevention and treatment of diseases.
Medicines are sub classification of the drugs. They produce good response and they are useful in order to diagnosis, prevention and treatment of diseases.
If taken in doses higher than those recommended, most of the drugs used as medicines are potential poisons.
Use of chemicals for treatment and cure of disease is called chemotherapy.
The treatment of disease by chemical compound which destroy the microorganisms without attacking the tissue of the human body is known as chemotherapy.
The compounds used are called chemotherapeutic agents.

Drugs and their Classification:
Drugs are chemicals of low molecular masses (~100-500u). They produce biological response by interacting with macromolecular targets. If the biological response is therapeutic and useful, these chemicals are called medicines. They are used in diagnosis, prevention and treatment of diseases.
Classification of Drugs
Drugs can be classified mainly on the basis of following criteria:
- On the basis of pharmacological effect
It is useful for doctors because it provides them whole range of drugs available for treatment of particular type of problem. Eg.- Analgesic – Shows pain killing effect. Antiseptic – Kill or arrest the growth of microorganisms.
- On the basis of drug action
It is based on action of drug on a particular biochemical process. Eg.- Histamines causes inflammation in the body and there are various ways in which action of histamines can be blocked. All antihistamines inhibit the action of the histamines.
- On the basis of chemical structure
It is based on chemical structure of the drug. Often drugs with common structural features, have similar pharmacological activity. Sulphonamides have common structural feature as shown above. Eg.
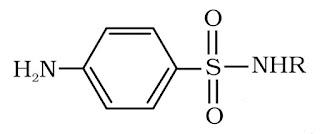
- On the basis of molecular targets
Drug possessing some common structural features, may have same mechanism of action on targets. These target molecules or drug targets are usually biomolecules such as carbohydrates, lipids, proteins and nucleic acids.
Drug-Target Interaction:
Macromolecules of biological origin perform various functions in the body. For example, Proteins which perform the role of biological catalyst in the body are called enzymes, those which are crucial to communication system in the body are called receptors. Carrier proteins carry polar molecules across the cell membrane. Nucleic acids have coded genetic information for the cell.
Enzymes as Drug Targets
- Catalytic action of enzymes
Enzymes perform two major functions :
- The first function of enzyme is to hold the substrate for chemical reaction. Enzymes have active sites, which hold the substrate molecule in a suitable position. The substrate can bind through enzyme by interactions such as ionic bonding, hydrogen bonding, van der Waals interaction or dipole-dipole interaction.
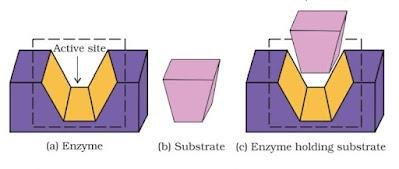
- The second function of enzyme is to provide functional groups that will attack the substrate and carry out chemical reaction.
- Drug-enzyme interaction
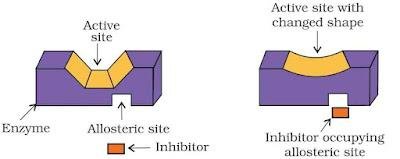
Enzyme inhibitors- Drugs can inhibit the activities of enzymes. They can block the binding site of the enzyme, thus prevent the binding of substrate or they can inhibit the catalytic activity of the enzyme. Drug can inhibit the attachment of substrate on active size of enzymes in following two ways :
- Competitive inhibitors : These are drugs which compete with natural substrate for their attachment on the active site of enzymes.
- Non-competitive inhibitors : These drugs do not bind to the enzyme’s active site, rather bind to a different site of enzyme called Allosteric site and changes the shape of active site in such a way that substrate can’t recognise it.
Receptors as Drug Targets
Receptors are proteins that are crucial to body’s communication process. Receptor proteins are embedded in cell membranes in such a way that their small part possessing active site projects out of the surface of the membrane and opens on the outside region of the cell membrane.
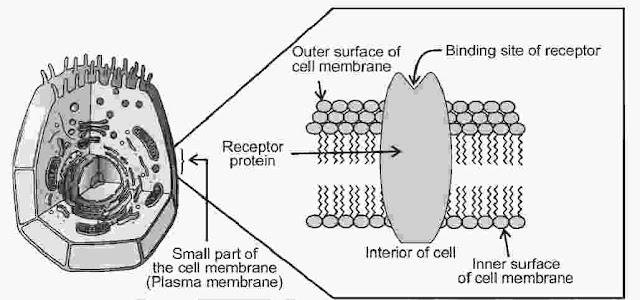
Chemical Messengers are the chemicals in the body, through which message between two neurons or that between neurons to muscles is communicated. They are received at binding sites of receptor proteins. To accommodate a messenger, shape of receptor site changes and brings about the transfer of message into the cell. Thus, chemical messenger give message to the cell without entering the cell.
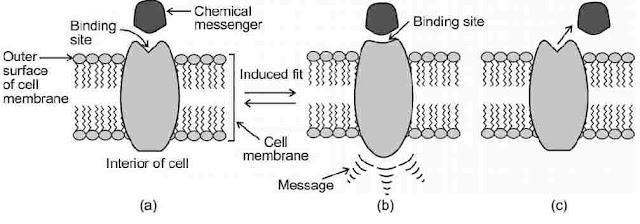
Drugs that bind to the receptor site and inhibit its natural function are called antagonists. These are useful when blocking of message is required. There are other types of drugs that mimic the natural messenger by switching on the receptor, these are called agonists. These are useful when there is lack of natural chemical messenger.
Therapeutic Action of Different Classes of Drugs:
Few important classes of drugs are :
- Antacids
Over production of acid in stomach causes pain and irritation and in severe cases ulcers are developed. Antacids such as sodium hydrogen carbonate or mixture of aluminium and magnesium hydroxide was used. But taking excess hydrogen carbonate makes the stomach alkaline and trigger the production of even more acid. Metal hydroxides are better antacids, as they are insoluble and do not increase the pH above neutrality.
- Antihistamines
Histamine is a potent vasodilator. It has various functions, like contraction of smooth muscles in the bronchi and gut and relaxing other muscles such as those in walls of fine blood vessels. Histamines are also responsible for nasal congestion associated with common cold and allergic response to pollen. Synthetic drug brompheniramine (Dimetapp) and terfenadine (Seldane) act as antihistamines.
- Neurologically Active Drugs
- Tranquilizers : They affect the message transfer mechanism from nerve to receptor. They are class of chemical compounds used for treatment of stress, mild or even severe mental diseases. They form essential component of sleeping pills. They relieve anxiety, stress, irritability or excitement by including sense of well being. Examples, Chlordiazepoxide, meprobamate are mild tranquilizers suitable for relieving tension.
- Analgesics : They reduce or abolish pain without causing impairment of consciousness, mental confusion, incoordination or paralysis or some other disturbances of nervous system. They are classified as :
- Non-narcotic (non-addictive) analgesics : Example- Aspirin, Paracetamol.
- Narcotic analgesics : Example- Morphine and its homologues like Heroin, Codeine etc.
- Antimicrobials
They destroy or prevent development or inhibit the pathogenic action of microbes such as bacteria (by antibacterial drug), fungi (by antifungal agents), virus (by antiviral agents) or other parasites (antiparasitic drugs) selectively.
Antibiotics : These are drugs used to cure infections because of low toxicity for humans and animals. Initially, they were classified as chemical substances produced by microorganisms, that inhibit the growth or even destroy other microorganisms.
- Antiseptic and Disinfectants
They are chemicals which either kill or prevent growth of microorganisms.
Antiseptics
They are applied to living tissues such as wounds, cuts, ulcers and diseased skin surfaces. They are not ingested like antibiotics. Examples are :
- Furacine, Soframycin.
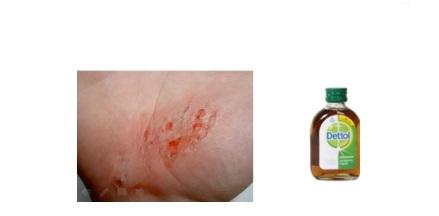
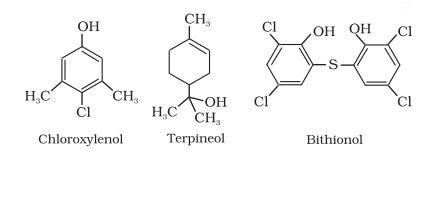
- Dettol, which is a mixture of chloroxylenol and terpineol.
- Bithionol, which is added to soaps to impart antiseptic properties.
- Iodine, which a powerful antiseptic, is a 2-3% solution in alcohol-water mixture, also known as tincture of iodine.
- Iodoform, which is also used as antiseptic for wounds.
Disinfectants
They are applied to inanimate objects such as floors, instruments, drainage system etc. Same substance can act as antiseptic as well as disinfectant by varying its concentration. For example, 0.2% solution of phenol is an antiseptic but 1% solution of phenol is disinfectant.
- Antifertility Drugs
They are used in direction of birth control and in family planning. Birth control pills essentially contains a mixture of synthetic estrogen and progesterone derivatives. Both of these compounds are hormones. Progesterone suppresses ovulation. Synthetic progesterone derivatives are more potent than progesterone. Example, Norethindrone a synthetic progesterone derivative, has antifertility action.
Chemicals in Food:
Chemicals are added to food for (i) their preservation, (ii) enhancing their appeal, and (iii) adding nutritive value in them. Main categories of food additives are as follows:
- Food colours
- Flavours and sweeteners
- Fat emulsifiers and stabilising agents
- Flour improvers- antistalling agents and bleaches
- Antioxidants Preservatives
- Nutritional supplements like minerals, vitamins and amino acids.
- Artificial Sweetening Agents
They are as sweet as sugar, but have no or less calories, whereas natural sweeteners e.g., sucrose add to calorie intake. It is used by a diabetic person and those who want to control their intake of calories. Ortho-sulphobenzimide, called saccharin is the first popular artificial sweetening agent, which is about 550 times as sweet as cane sugar. It is excreted from the body in urine unchanged. It is harmless and appears to be entirely inert.
Other examples are :
- Aspartame: It is the most successful and widely used sweetener. It is roughly 100 times as sweet as cane sugar. It is methyl ester of dipeptide formed from aspartic acid and phenylalanine. As it is unstable at cooking temperature, its use is limited to cold foods and soft drinks.
- Alitame : It is a high potency sweetener, more stable than aspartame. But control of sweetness of food is difficult while using it.
- Sucralose : It is a trichloro derivative of sucrose. Its appearance and taste are like sugar. It is stable at cooking temperature and does not provide calories.
Food Preservatives
They prevent spoilage of food due to microbial growth. Commonly used preservatives are table salt, sugar, vegetable oils, sodium benzoate (C6H5COONa), salts of sorbic acid and propanoic acid. Sodium benzoate is used in limited quantities and is metabolised in the body.
Cleansing Agents
They improve cleansing properties of water and help in removal of fats which bind other materials to the fabric or skin. They include-
Soaps: Soaps used for cleaning purpose are sodium or potassium salts of long chain fatty acids e.g., stearic, oleic and palmitic acids. Soaps containing sodium salts are formed by heating fat (i.e., glyceryl ester of fatty acid) with aqueous sodium hydroxide solution. This reaction is known as saponification.
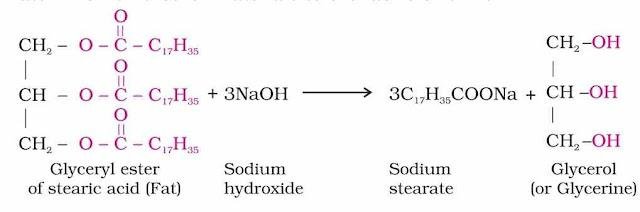
In this reaction, esters of fatty acids are hydrolysed and soap obtained remains in colloidal form. It is precipitated from solution by adding sodium chloride. The solution left after removing soap contains glycerol and can be recovered by fractional distillation.
Types of Soaps
- Toilet Soaps : They are prepared by using better grades of fats and oils and excess of alkali is removed. Colour and perfumes are added to make these more attractive.
- Transparent Soaps : They are made by dissolving the soap in ethanol and then evaporating the excess solvent.
- Medicated Soaps : Substances of medicinal values are added.
- Shaving Soaps : Contain glycerol to prevent rapid drying. A gum called, rosin is added while making, it forms sodium rosinate which lathers well.
- Laundry Soaps : These contains fillers like sodium rosinate, sodium silicate, borax and sodium carbonate.
Why soaps do not work in hard water?
As we know that hard water contains calcium and magnesium ions which forms insoluble salts with soaps, that gets separated as scum in water.
2C17H35COONa + CaCl2 ⟶ 2NaCl + (C17H35COO)2Ca
In fact, the scum so separated offers hinderance to soap action as the precipitate adheres onto the fabric as gummy mass. Also, the hair washed with hard water looks dull because of this sticky precipitate. Similarly, dyes do not absorb evenly on cloth washed with soap using hard water.
Limitations of Detergents
The problem of using detergents is that if their hydrocarbon chain is highly branched, then bacteria cannot degrade this easily and their slow degradation leads to their accumulation. Effluents containing such detergents reach rivers, ponds etc. and persist in water even after sewage treatment. This cause foaming in rivers, ponds and streams which pollutes the water.
Now a days, the branching of hydrocarbon chain is controlled and kept to the minimum. Unbranched chains can be biodegraded more easily and hence pollution is prevented.
Antimicrobials
Antimicrobials
Diseases in human beings and animals may be caused by a variety of microorganisms such as bacteria, virus, fungi and other pathogens.
An antimicrobial tends to destroy/prevent development or inhibit the pathogenic action of microbes such as bacteria (antibacterial drugs), fungi (antifungal agents), virus (antiviral agents), or other parasites (antiparasitic drugs) selectively.
Antibiotics, antiseptics and disinfectants are antimicrobial drugs.
Antibiotics
Antibiotics are used as drugs to treat infections because of their low toxicity for humans and animals.
An antibiotic is a substance produced wholly or partly by chemical synthesis, which in low concentrations inhibits the growth or destroys microorganisms by intervening in their metabolic processes.
In order to find chemicals this will affect the invading bacteria and not the host.
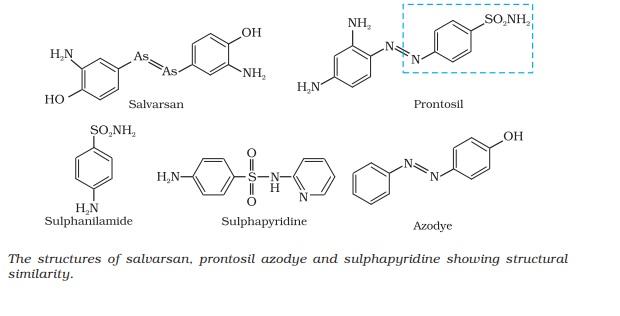
Paul Ehrlich, a German bacteriologist, conceived this idea. He investigated arsenic based structures in order to produce less toxic substances for the treatment of syphilis.
He developed the medicine, arsphenamine, known as salvarsan.
Although salvarsan is toxic to human beings, its effect on the bacteria, spirochete, which causes syphilis, is much greater than on human beings.
He noted that there is similarity in structures of salvarsan and azodyes. The –As = As– linkage present in arsphenamine resembles the –N = N – linkage present in azodyes in the sense that arsenic atom is present in place of nitrogen.
In 1932, he succeeded in preparing the first effective antibacterial agent, prontosil, which resembles in structure to the compound, salvarsan. Soon it was discovered that in the body prontosil is converted to a compound called sulphanilamide, which is the real active compound. Thus the sulpha drugs were discovered.
A large range of sulphonamide analogues was synthesised. One of the most effective is sulphapyridine.
Despite the success of sulphonamides, the real revolution in antibacterial therapy began with the discovery of Alexander Fleming in 1929, of the antibacterial properties of a Penicillium fungus.
Antibiotics have either cidal (killing) effect or a static (inhibitory) effect on microbes.
A few examples of the two types of antibiotics are as follows:
Bactericidal Bacteriostatic
Penicillin Erythromycin
Aminoglycosides Tetracycline
Ofloxacin Chloramphenicol
Antibiotics which kill or inhibit a wide range of Gram-positive and Gram-negative bacteria are said to be broad spectrum antibiotics.
Those are effective mainly against Gram-positive or Gram-negative bacteria are narrow spectrum antibiotics.
If effective against a single organism or disease, they are referred to as limited spectrum antibiotics.
Penicillin G has a narrow spectrum. Ampicillin and Amoxicillin are synthetic modifications of penicillin’s. These have broad spectrum.
It is absolutely essential to test the patients for sensitivity (allergy) to penicillin before it is administered.
In India, penicillin is manufactured at the Hindustan Antibiotics in Pimpri and in private sector industry.
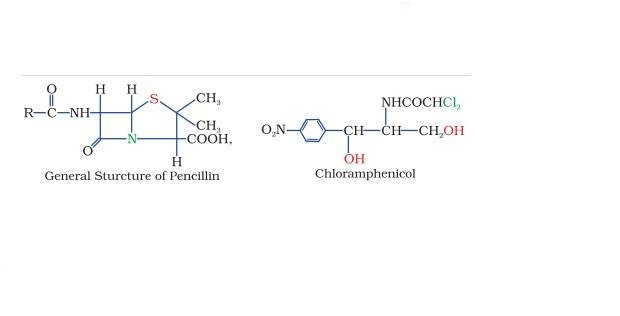
Chloramphenicol
Chloramphenicol, isolated in 1947, is a broad spectrum antibiotic.
It is rapidly absorbed from the gastrointestinal tract and hence can be given orally in case of typhoid, dysentery, and acute fever, certain form of urinary infections, meningitis and pneumonia.
Vancomycin and ofloxacin are the other important broad spectrum antibiotics.
The antibiotic dysidazirine is supposed to be toxic towards certain strains of cancer cells.
Antiseptics & disinfectants
- Antiseptics are applied to the living tissues such as wounds, cuts, ulcers and diseased skin surfaces.
- Examples are furacine, soframicine, etc. These are not ingested like antibiotics.
- Commonly used antiseptic, Dettol is a mixture of chloroxylenol and terpineol.
- Bithionol (the compound is also called bithionol) is added to soaps to impart antiseptic properties.
- Iodine is a powerful antiseptic. Its 2-3 per cent solution in alcohol water mixture is known as tincture of iodine.
- It is applied on wounds.
- Iodoform (CHI3) is also used as an antiseptic for wounds.
- Boric acid in dilute aqueous solution is weak antiseptic for eyes.
Disinfectants
- They are applied to inanimate objects such as floors, drainage system, instruments, etc. Same substances can act as an antiseptic as well as disinfectant by varying the concentration.
- For example, 0.2 per cent solution of phenol is an antiseptic while its one percent solution is disinfectant.
- Chlorine in the concentration of 0.2 to 0.4 ppm in aqueous solution and sulphur dioxide in very low concentrations, are disinfectants.
Antifertility Drugs
- These are the chemical substances which are used to control pregnancy.
- They are also called as oral contraceptives.
- Birth control pills essentially contain a mixture of synthetic estrogen and progesterone derivatives. Both of these compounds are hormones.
- It is known that progesterone suppresses ovulation.
- Synthetic progesterone derivatives are more potent than progesterone.
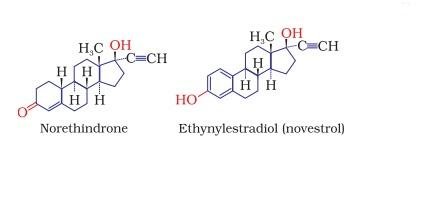
- Norethindrone is an example of synthetic progesterone derivative most widely used as antifertility drug. The estrogen derivative which is used in combination with progesterone derivative is ethynylestradiol (novestrol).
NCERT Solutions for Class 12 Chemistry chapter wise
- Chapter 1 The Solid State
- Chapter 2 Solutions
- Chapter 3 Electrochemistry
- Chapter 4 Chemical Kinetics
- Chapter 5 Surface Chemistry
- Chapter 6 General Principles and Processes of Isolation of Elements
- Chapter 7 The p Block Elements
- Chapter 8 The d and f Block Elements
- Chapter 9 Coordination Compounds
- Chapter 10 Haloalkanes and Haloarenes
- Chapter 11 Alcohols Phenols and Ethers
- Chapter 12 Aldehydes Ketones and Carboxylic Acids
- Chapter 13 Amines
- Chapter 14 Biomolecules
- Chapter 15 Polymers
- Chapter 16 Chemistry in Everyday Life
